AbstractMany species of marine algae have life cycles that involve multiple separate, free-living phases that frequently differ in ploidy levels. These complex life cycles have received increasing scientific attention over the past few decades, due to their usefulness for both ecological and evolutionary studies. I present a synthesis of our current knowledge of the ecological functioning and evolutionary implications of the isomorphic, biphasic life cycles commonly found in many species of marine algae. There are both costs and benefits to life cycles with 2 morphologically similar but separate, free-living phases that differ in ploidy levels (haploids and diploids). Evolutionary theory predicts that the existence of subtle yet important differences between the phases may be what allows these life cycles to be maintained. Different phases of the same species can vary in abundance, in demographic parameters such as mortality and fecundity, in their physiology, and in their resistance to herbivory.
In the life cycle of Polysiphonia, and many other red algae, there are separate male and female gametophytes, carposporophytes that develop on the female gametophytes, and separate tetrasporophytes. The life cycles of diatoms, which are diploid, are also unique. Most algae have two recognisable phases - the sporophyte and the gametophyte. The main types of algal life cycles are exhibited by green algae. (Red algae have even more complicated life cycles!) Some Green algae are unicellular and demonstrate the simplest possible life cycles.
Some taxonomic groups within the red algae have received significant attention toward these issues, while our knowledge of these properties for brown and green algae remains limited. IntroductionComplex life cycles, which involve the existence of more than one stage or phase for a species, are commonly found in a wide variety of marine species, including primary producers, herbivores, and predators. Species with such life cycles face unique ecological and evolutionary constraints, as each and every stage/phase must be able to survive and/or reproduce for the species to persist. Individuals may undergo dramatic developmental and morphological changes as they age (for example from larval to adult stages,; ). Alternatively, other species with complex life cycles such as many algae and plants must cycle through multiple ploidy stages. In either case, these stages may occupy similar or different ecological niches and face varying environmental constraints (see review in ). Because these stages are obligately linked to each other, impacts or effects on one stage may cascade through the species' entire life cycle (; ).An array of multiphasic lifecycles can be found in marine algae.
Complex life cycles in marine algae are widely distributed across different taxa, including the three groups that contain marine macroalgae: the green (Chlorophyta), the brown (Phaeophyceae), and the red (Rhodophyta) seaweeds. This review is intended to synthesize our existing ecological knowledge of species with biphasic life cycles and provide a perspective of the evolution and maintenance of these life cycles. In particular, I focus on marine macroalgal species with isomorphic biphasic life cycles and our increased understanding of the ecological similarities and differences between apparently identical phases. Algal life cyclesMost algal life cycles can be separated into 1 of the 3 general categories: gametic (diplontic), zygotic (haplontic), and biphasic (sporic/haplo-diplontic; ). Species with gametic life cycles consist primarily of diploid organisms, with meiosis occurring at gamete formation; gametes are the only haploid phase and rapidly fuse to create a new diploid zygote.
This is perhaps the most familiar life cycle, as many animals (including humans) have some type of this general life cycle. Species with zygotic life cycles consist primarily of haploid organisms; the only diploid stage is a zygote (from the fusion of two haploid gametes), which rapidly undergoes meiosis to produce new haploid spores or cells.
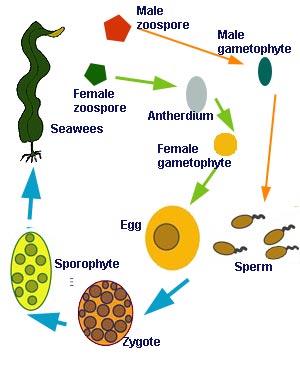
The haploid cells then grow into new gametophytes that, when mature, produce haploid gametes.This review focuses on the biphasic life cycle, which is the most complex of the three. It generally involves an obligate cycling between separate, free-living, and independent haploid (gametophyte) and diploid (sporophyte) phases. Haploid gametes are produced from mature, multicellular haploid gametophytes and released into the water column.
Two gametes (usually male and female, but for some green algae the gametes are referred to as + and −) then fuse to create a diploid zygote, which settles and then grows into a mature, multicellular diploid sporophyte. Meiosis occurs when haploid spores are formed by the sporophyte; these spores are released into the water column, settle, and then grow into new gametophytes. Although gametophytes are usually dioecious (separate sexes), as shown in, they are monoecious in some species. This distinction can have important implications for the relative abundance of the 2 phases (see Theoretical predictions). (a)The biphasic haploid–diploid life cycle. (b) The triphasic red algal life cycle in the Florideophyceae. See Algal life cycles for a complete description.Most species of red algae have a modified biphasic life cycle, which includes a third, short-lived diploid carposporophyte stage.
This diploid stage is formed by the fusion of haploid gametes on the surface of the female gametophyte thallus (as opposed to both gametes being released and fusing in the water column). This carposporophyte lives attached to the female gametophyte, acquiring nutrients from it , and quickly becomes a mass of diploid spores. These diploid carpospores are released into the water column, settle, and become new free-living tetrasporophytes. Tetrasporophytes, when mature, produce haploid tetraspores via meiosis (the prefix tetra is because the spores are packaged in groups of fours). These tetraspores are released into the water column, settle, and grow into male and female haploid gametophytes. This triphasic life cycle is thought to have evolved as a mechanism of increasing reproductive output , because red algae are unique among algal taxa in lacking flagella on any of their reproductive cells (and thus cannot swim to find a mate).
Because these red algal species have life cycles with two independent, free-living phases (although they are technically called “triphasic”), in this review they will be considered together with the other algal species (browns and greens) that have biphasic life cycles. The terms sporophyte and tetrasporophyte will be used to refer to the diploid free-living phase of the life cycle. Life cycle modificationsThere are a variety of functional constraints that can impact species with biphasic life cycles. First, and perhaps the most important, are the effects of apomixis (asexual looping) that may allow an algal species to circumvent a part of its life cycle (for reviews of this process in red algae, see Maggs 1988;; ).
In some populations, a gametophyte may thus be able to produce new gametophytes without going through the sporophyte phase. The reverse may also occur, with sporophytes directly producing new sporophytes (; ). Other life cycle variations include individuals that produce both carpospores and tetraspores on the same thallus , species that reproduce without ever producing males , and species that may be entirely asexual and never exhibit a full life cycle (Maggs 1998; ). Heteromorphy versus isomorphyAnother functional constraint that algae with biphasic life cycles face is the separate life history strategies of the gametophyte and sporophyte phases. These 2 free-living, independent phases range from appearing morphologically distinct from one another (heteromorphy) to seemingly virtually identical to each other (isomorphy), depending upon the species. Many studies have observed this and questioned the relative advantages or drawbacks to either strategy (for example; ).
Advantages of heteromorphic life cycles include the 2 phases' abilities to exploit different ecological niches by being adapted to environments that differ in terms of temperature, light levels, or herbivore abundance (;; ). For example, all kelps (large brown algae) have heteromorphic life cycles with a large, macroscopic sporophyte and a tiny, microscopic gametophyte. The microscopic gametophytes of the kelp Lessonia nigrescens are more heavily grazed, but less impacted by wave action, than slightly larger, very juvenile microscopic sporophytes.

Other species, such as the red alga Mastocarpus and the brown alga Scytosiphon, have 2 macroscopic phases; in these genera one phase is upright (foliose) and the other phase is crustose (; ). The slower-growing crustose phase is frequently much more resistant to herbivory and may provide a refuge for the species when herbivore pressure is high (;; ).Understanding the functional properties of species with isomorphic life cycles can prove to be more challenging (; ). Haploid and diploid phases of these species appear virtually identical (especially when nonreproductive) and frequently overlap spatially and temporally in their habitats. However, a growing body of literature provides evidence for a range of ecologically significant variation between isomorphic phases (see Ecological differences sections). Isomorphic life cycles exist in several distantly related groups of brown, green, and red algae (;; C. Thornber unpublished data).
Evolutionary implicationsWhy do biphasic, isomorphic life cycles exist in nature? An array of studies have used a combination of evolutionary and genetic approaches to address this question (see reviews by; ). There are costs and benefits to both ploidy states; diploid advantages include the ability to mask mutations and adapt more quickly to environmental variation. Haploid advantages, by comparison, may include lower mutational loads and reduced nutrient requirements.
Thus, for a particular set of conditions, a zygotic or gametic life cycle may be favored or predicted to evolve over time (;; ). However, haploid–diploid life cycles have been predicted to evolve in some recent genetic models (;; ). The stability of these isomorphic biphasic life cycles can be promoted by slight, but ecologically significant, differences in demographic parameters between the phases. Theoretical predictionsDetermining the ratio of isomorphic gametophytes to sporophytes in field populations is of paramount importance to interpreting field data. Due to this, theoretical population dynamic models that explore the relative abundance of the 2 phases have received increasing attention over the past 2 decades. Early models such as those by and were the first to provide a theoretical basis for gametophyte: sporophyte ratios. Subsequent models explored both general conditions for ratios in isomorphic algae (; ) as well as specific cases incorporating a more detailed knowledge of a species' biology (; Engel and others 2001; Fierst and others 2005).
In general, if the species must obligately cycle (no apomixis) between 2 phases that are ecologically equivalent, that is, they have equal per capita demographic rates, and the species is dioecious (separate male and female gametophytes), then a ratio of √2 gametophytes: 1 sporophyte is expected at equilibrium. If a species is monoecious, a ratio of 1:1 is expected. However, this ratio may shift if there are differences in factors such as survival, spore recruitment, coalescence, fecundity, fertilization success, and disease (among other factors). For example, if one phase has a much higher per capita fecundity rate, this should result in a higher proportion of the alternate phase at equilibrium.
Thus, simply because one phase is numerically more abundant should not imply that it is ecologically “superior” or a better competitor than the other phase. If apomixis is present for either phase, these demographic proportions could also shift substantially. Population patternsAn area that has received considerable attention in studies of isomorphic biphasic life cycles is the relative balance between the 2 phases in field populations. Understanding this balance may yield insight into the functioning of complex life cycles, as each phase produces the other (except in cases of apomixis) through reproduction.
Some of the first field studies included surveys of only reproductive sporophytes and reproductive (female) gametophytes, which can be the easiest to distinguish visually in the field. This may, however, provide an incomplete estimate of the total distribution of phases, as neither reproductive males nor nonreproductive individuals were included. Regardless of such biases, surveys of this type reported a variety of gametophyte–sporophyte ratios, ranging from gametophyte predominance for Mazzaella laminarioides (; ) and 2 unidentified species of Gracilaria , to equal numbers of the phases for Gracilaria cuneata and Gracilaria gracilis to sporophyte predominance in Gracilaria domingensis , Mazzaella flaccida , Mazzaella splendens , and Padina japonica. Additional surveys are summarized in.Surveys of all individuals in isomorphic populations are more difficult to conduct, but techniques such as the resorcinol test and spectroscopy enable the identification of nonreproductive individuals for some species. These latter surveys have found a variety of distributions. A consistent numerical dominance of gametophytes was determined for Cladophoropsis membranacea , Chondrus crispus (; ), and Mazzaella capensis, M.
Laminarioides, and M. Splendens (; ). A numerical dominance of sporophytes was determined for Dictyota binghamiae , Mazzaella splendens , and Padina sanctae-crucis (Little 1972), and equal numbers of the 2 phases were documented for populations of Chondrus crispus. Increasingly common are studies that document spatial and/or temporal fluctuations between a numerical dominance of gametophytes and sporophytes, including Gracilaria gracilis , Mazzaella capensis , Mazzaella oregona , Mazzaella splendens (;; ), and Sarcothalia crispata. These studies and others (; ) that have examined populations over a range of temporal and/or spatial scales allow for the tracking of longer-term (or broader-scale) trends in the population dynamics of the 2 phases. The accumulation of data on population ratios for species, such as Mazzaella splendens, allows for a much more comprehensive understanding of how these phases may fluctuate.
However, these studies represent a small minority of the species that have isomorphic life cycles; for most species, little if any data exist on their population structure. A recent summary table of most published red algal gametophyte–tetrasporophyte surveys was compiled by and indicates a general trend toward gametophyte dominance in the order Gigartinales and tetrasporophyte dominance in the orders Gracilariales and Ceramiales. Ecological differences: demographic ratesDifferences in per capita demographic parameters (broadly grouped into mortality and fecundity rates) between gametophyte and sporophyte phases may help explain the variability in gametophyte/sporophyte ratios in the field. Studies of these rates are crucial to understanding why 1 phase may be more (or less) abundant in field populations than predicted.For the purposes of this review, per capita fecundity is defined as the number of carpospores produced on 1 female gametophyte (for red algae) and the number of tetraspores produced by 1 tetrasporophyte.
Several studies found no difference in per capita fecundity between the phases for Hypnea cervicornis and Hypnea chordacea , Mazzaella laminarioides , Mazzaella splendens , and Plocamium cartilagineum. This was true even though total diploid carpospore production (by gametophytes) was higher than total haploid tetraspore production (by tetrasporophytes) in a Mazzaella laminarioides field site. However, tetrasporophyte fecundity was significantly higher than gametophyte fecundity in Gracilaria gracilis and Mazzaella flaccida , while gametophyte fecundity was higher than tetrasporophyte fecundity for Gelidium sesquipedale. Peaks in reproductive output between phases may also vary seasonally; this was shown for field populations of Gelidium sesquipedale , but no seasonal shifts between the phases were seen for Mazzaella flaccida. Differences in fecundity between the phases can also be spatially variable; tetrasporophytes of the subtidal species Ptilota serrata were reproductive at deeper depths than gametophytes, but reproductive depths for the 2 phases were equivalent for Callophyllis cristata, Membranoptera alata, and Phycodrys rubens.At the microscopic stage, species-specific differences can and do exist between tetraspore and carpospore survival, dispersal, and settlement rates.
Haploid tetraspores of Chondracanthus chamissoi had higher settlement and germination rates. In contrast, haploid tetraspores and newly settled haploid germlings of Gracilaria gracilis had higher mortality rates, but tetraspores had the potential for longer dispersal distances (, ). Haploid tetraspores of Gracilaria pacifica had increased survival rates in winter. Spore settlement rates were greater for carpospores of Gelidium sesquipedale at lower temperatures, while tetraspore settlement rates were greater at higher temperatures.Per capita mortality rates of larger (macroscopic) individuals were equal for the 2 phases for Gracilaria gracilis (; ), Mazzaella flaccida and Mazzaella laminarioides , and Mazzaella splendens. Ecological differences: physiology/growth/herbivoryDifferences in demographic rates between isomorphic gametophytes and sporophytes may result from a broad suite of factors such as differences in size, growth rates, light and temperature optima, and/or herbivore selectivity.
The significance of these functional differences in phases of isomorphic species is becoming increasingly recognized. Some isomorphic species may differ morphologically: tetrasporophytes of Gelidium sesquipedale were larger and less branched than their gametophyte counterparts , while branching patterns differed between phases for Ceramium codicola. Size or structural differences between phases could impact mortality rates (if, for instance, larger individuals are more likely to be removed from their substrate in large wave events) and/or fecundity rates (larger individuals may have more tissue to support reproductive structures).Some studies have examined differences in growth rates between the two phases. The growth rates of both phases were the same for Mazzaella flaccida (C. Thornber unpublished data) and Mazzaella laminarioides. Adult gametophytes grew more rapidly than tetrasporophytes of Chondracanthus chamissoi and Iridaea ciliolata. Sporophyte growth rates were faster for Caloglossa leprieurii and post-settlement stages of Chondracanthus chamissoi.
Tetrasporophytes of Gelidium sesquipedale were significantly better at vegetatively propagating than are gametophytes.Rates of photosynthesis and respiration, where measured, tend to be similar for the 2 phases. This has been shown in research involving Gelidium canariensis , Gelidium sesquipedale , and Endocladia muricata (except in some cases of higher photosynthesis rates in tetrasporophytes after prolonged emergence in air).Caloric and protein content were similar in the 2 phases of Gelidium canariensis and Polycavernosa debilis , and protein content and chemical defenses were similar in Dictyota ciliolata. Not all tissue properties are the same; however, the percentage organic content was higher in Mazzaella flaccida female gametophyte reproductive tissue than sporophyte tissue (Thornber and others 2006). Also, several species in the Gigartinaceae differ in their carrageenan content between the 2 phases (; ); gametophytes have kappa-type carrageenans, while tetrasporophytes have lambda-type carrageenans.
Carrageenans are cell wall phycocolloids, and differences in these compounds may impact the ability to tolerate wave exposure (; ).Phases may differ in their ability to withstand wave forces, which is of particular importance to intertidal species. Chondrus crispus gametophytes were stronger than tetrasporophytes, but both phases were equally likely to be dislodged by wave action. Gametophytes of Padina japonica were more susceptible to tearing in increased water flow than sporophytes.Herbivores may or may not distinguish between isomorphic phases; if differences exist, they may be reflected either in mortality rates (if an herbivore eats entire individuals or enough tissue to lower the probability of survival) or in fecundity rates (if an herbivore selectively grazes and digests reproductive tissue of a particular phase). Herbivores eating Mazzaella flaccida selectively grazed mature female gametophyte (with attached carposporophyte) tissues over nonreproductive gametophyte or tetrasporophyte tissue (Thornber and others in press); some herbivores demonstrated the same preference for Mazzaella laminarioides (; ). However, no herbivore preference was found between the isomorphic phases of Dictyota ciliolata or Polycavernosa debilis (Littler and others 1987).There are many ways in which isomorphic phases may differ significantly from each other; the studies summarized above have examined a variety of these ecological parameters. Overall, there is no consistent trend of one phase outperforming another; instead, significant variability exists both among species and among ecological parameters.
Genetic population structureStudies on the genetic variability and genetic differentiation of isomorphic phases have become increasingly common during the past 2 decades (see review by ). One of the first studies to examine this was, who found a higher genetic variability in diploid subpopulations than haploid subpopulations of Gelidium arbuscula; subsequent analyses indicated that these populations mainly reproduce asexually. There was twice as much genetic differentiation in Gracilaria gracilis among gametophytes than among tetrasporophytes. In one of the few studies that examined isomorphic phases in green algae, found no differences in genetic diversity between the 2 phases of Cladophoropsis membranacea. Intra-phasic differences in the physiology of different identified haplotypes can also be important; growth rates of the red alga Caloglossa leprieurii differed by both haplotype and phase (tetrasporophytes grew faster than gametophytes; ).
Future directionsOur knowledge about the population dynamics and functional properties of isomorphic phases is rapidly increasing. However, nearly all of the published studies on gametophyte: sporophyte ratios (where both reproductive and nonreproductive individuals were surveyed), and ecological differences between the phases, focus on red algae, in particular on species within the orders Gigartinales ( Mazzaella), Gracilariales ( Gracilaria), Ceramiales and Gelidiales; see summary table in Fierst and others (2005).
